It's all in the name

In science there are two different types of matter. They are called "pure substances" and "mixtures". Pure substances are essentially that; pure. That means that they can't be separated by any means. All pure substances have their own set of chemical and physical properties. This means that every sample of a pure substance you take will be the same. Lets say you take some good old liquid water and separate it out into three identical glass jars. All of these samples will have the same properties. No matter how you look at it they will all still be clear, odorless, liquids. They will all boil at 100 degrees celsius and (if ice) will all melt at 0 degrees celsius. That is the beauty of a pure substance.
elements

If we get even further into it, there are two types of pure substances! One is called and element, and the other a compound. An element is a pure substance in its "purest state". That means if something is an element, it can not be broken apart into anything else. You can find elements on the periodic table of elements. Examples of elements would be oxygen, gold, and silver.
Compounds
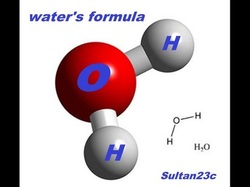
Compounds are made of two or more elements. You've probably seen this before, you know... H20? Well believe it or not, water is a compound substance. There are two hydrogen atoms to one oxygen atom. As long as this ratio of 2:1 stays the same per molecule, you can have as much water as you want!
How can i tell the difference?
The easiest way to tell the difference between a compound substance and an element is in the chemical formula. If a chemical formula only has one element, for example O2 (oxygen) you know that it is an element. If it has two or more elements such as H2O or CO2 we know that it is a compound substance.
"In learning you will teach, and in teaching you will learn." Phil Collins
